Chinese vaccine experts share production know-how in Pakistan
As many as 120,000 vaccines doses have recently been produced at Pakistan's National Institute of Health (NIH)
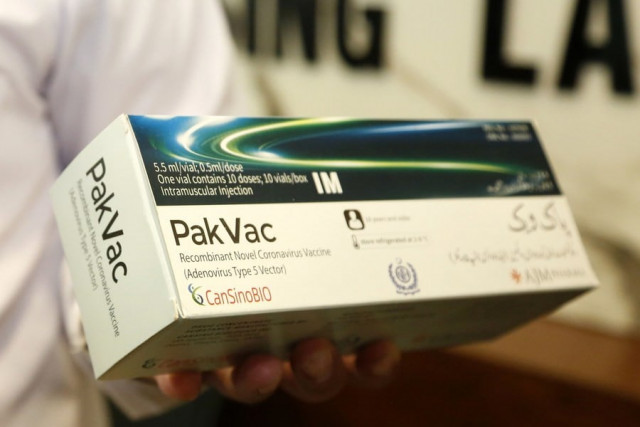
Pakistan's pandemic efforts have received a "booster shot" as the first batch of locally-produced Covid-19 vaccines is rolling off the line here at a national lab.
As many as 120,000 doses have recently been produced at Pakistan's National Institute of Health (NIH) under rigorous quality control with tech support from Chinese pharmaceutical firm CanSino.
Local health authorities welcomed the vaccines as a major landmark in Pakistan's efforts to attaining vaccine self-sufficiency, a much-needed, yet challenging task that Pakistan had fervently hoped to fulfil within months.
Also read: Homemade ‘PakVac’ launched to boost vaccination
"The cooperation between the two countries also underscores the philosophy that the Covid-19 vaccine is a global good," said Special Assistant to the Pakistani Prime Minister on Health Faisal Sultan at the production ceremony.
Magic in an empty room
CanSino Biologics Vice President Xin Chunlin admitted it initially came to him that the first batch of local production "wouldn't be a tough assignment," given the NIH's rich experience in vaccine manufacturing.
The reality proved the opposite -- making recombinant Covid-19 vaccines is a brand-new practice for the South Asian country, key aspects of which, including facilities and quality control standards, had to start from scratch, said Xin, who also leads a taskforce with members from both countries.
The task was further challenged by the global supply shortage including key equipment for a lab, chemicals necessary for the production, and subsequent reliance on some manual work.
"It's like we were asked to start our experiments in an empty room," Xin recalled.
Despite all these challenges, the team created a facility that can conduct analyses such as western-blotting essential to the production process.
Working alongside Chinese counterparts has enabled Pakistani members to gain hands-on experience in key quality control techniques such as virus particle testing, said Rafique Ahmed Channa, a Pakistani with more than 30 years of experience in the field.
Also read: First batch of another million doses of Sinopharm vaccine reaches Pakistan
The team also overcame ingredient shortages by finding best suited substitutes via rounds of trial-and-error.
"It's like wading the river by groping for stones. We shared our own experiences so as to find the best solution," said Xin.
MAKING KNOWLEDGE MORE INCLUSIVE
In the eyes of Pakistani experts, each and every attempt will add up to upgrading the country's health infrastructure and final fulfilment of self-sufficiency.
The localisation came as another first-time experience for the South Asian country in Covid-19 vaccine technology, after a successful phase III clinical trial of the vaccine under the cooperation between the NIH and CanSino.
So far, the Drug Regulatory Authority of Pakistan has approved three types of Chinese vaccines developed by pharmaceutical companies Sinovac, CanSino and Sinopharm, for emergency use.
Technology transfer enabled by bilateral cooperation has helped Pakistan to stride forward in achieving vaccine production capacity, said Ghazala Parveen, chief of the Biological Production Division at the NIH.
"The technology transfer is extremely crucial for the people of Pakistan. It helps us save money rather than buying vaccines from other countries," said Saba Haq, research official at the NIH.
New data from the Bloomberg Vaccine Tracker shows that the least wealthy 37 places worldwide covering more than a tenth of the world population have only received one percent of all doses.
Yet countries like China have been striving to expand the global supply of the life-saving shots, closing the widening gap between rich and poor nations.
From his experience, Xin said the most urgent task is to ensure the supply chain of vaccine production. "Take syringes as an example. It may not be the focal point when you talk about vaccine patents. But it can be the thing that slows down the delivery of the vaccines," he said.
More work needs to be done in making vaccine production technology transferable to developing countries, he added.
Seeing the upgraded infrastructure and better-trained manpower, Parveen said it gave her greater confidence that her country can now better handle infectious diseases.
"If another pandemic in the future will arrive, we know how to control it," she said.



















COMMENTS
Comments are moderated and generally will be posted if they are on-topic and not abusive.
For more information, please see our Comments FAQ