Painkillers like Advil or Motrin cause increased risk of heart attack, stroke
FDA says people should think carefully about taking these drugs
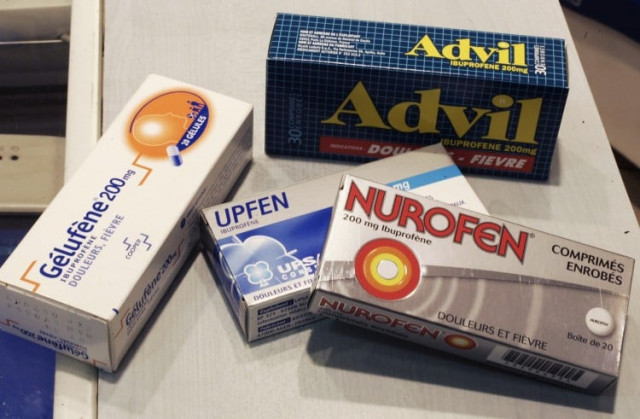
PHOTO: AFP
People should think carefully about taking these drugs, both over-the-counter versions and prescription pills, the FDA says. It's asking manufacturers to change the labels.
While the previous labels did include a warning that said such drugs “may cause” increased risk of heart attack and stroke, new evidence has suggested a strong link between increased risk of heart failure from such drugs which are consumed by millions worldwide.
Non-aspirin painkillers classified under NSAIDs are an important and oft relied upon source of treatment for many incapacitating illnesses including osteoarthritis, rheumatoid arthritis, gout and other rheumatological and painful disorders. They include ibuprofen, sold under brand names like Advil or Motrin; naproxen (Aleve), as well as prescription arthritis drugs known as COX-2 inhibitors, such as Celebrex.
Read:Improving healthcare: A new way to practice medicine in Pakistan
Both over-the-counter drugs and prescription pills should be approached with caution warns the FDA which is asking manufacturers to change their labels to include the notice.
Over-the-counter drugs are commonly used by people to relieve symptoms of cold and flu, headaches, toothaches, backaches, muscular aches, tendonitis, strains, sprains and menstrual cramps.
Read:Painkiller abuse ups risk of depression
"In the coming months, the FDA will request that manufacturers update the existing cardiovascular risk information in Drug Facts labels for over-the-counter (OTC) non-aspirin NSAIDs. Consumers and health care professionals should remain alert for the development of heart- and stroke-related symptoms throughout the time a consumer takes any NSAID," FDA said.
“Everyone may be at risk, even people without an underlying risk for cardiovascular disease,” Dr Judy Racoosin, the deputy director of the FDA’s Division of Anesthesia, Analgesia and Addiction Products said explaining the alteration.
This doesn't mean people should just stop taking NSAIDS, FDA said.
"Take the lowest effective dose for the shortest amount of time possible," said FDA's Dr. Karen Mahoney.












1725429132-0/Untitled-design-(12)1725429132-0-208x130.webp)






COMMENTS
Comments are moderated and generally will be posted if they are on-topic and not abusive.
For more information, please see our Comments FAQ